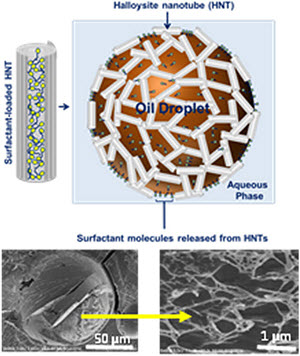
Cover of the September 2016 Oceanography Magazine, Volume 29, Number 3
7th year of the largest coordinated research endeavor around an ocean event.
The 2010 Deepwater Horizon oil spill and subsequent response efforts raised concerns about impacts on the Gulf of Mexico’s ocean and coastal environments. The Gulf of Mexico Research Initiative (GoMRI), in response to the spill, initiated an unprecedented 10-year scientific research program funded by BP. Seven years into the program, we know more than ever before about the Gulf’s complex environment, dynamic processes, and response to stressors.
Oceanography magazine dedicated a special issue to this research, GoMRI: Deepwater Horizon Oil Spill and Ecosystem Science, and below are highlights from 13 papers it featured.*
WHERE OIL WENT
Surface oil covered a cumulative area of 149,000 km2 in the northeastern Gulf. Wind and currents transported surface slicks towards land, affecting approximately 1,800-2,100 km of shoreline, a third of which were moderately to heavily oiled including 1,075 km in Louisiana. Macondo oil was visually evident at the edge of Louisiana marshes and up to 10 m inland.
Subsea oil and gas rose through the water column and formed an underwater oil plume that covered an area of approximately 930 km2 and made direct contact with continental slope sediments. A significant proportion of surface oil returned to the deep seafloor primarily through an extensive marine oil snow sedimentation event known as a “dirty blizzard,” forming a 0.5-1.2 cm thick floc layer.
Cleanup efforts removed oil from 73% of beaches affected by the spill, but residual oil remained as surface residue balls (SRBs), submerged oil mats, and in marsh plants and sediment, and is subject to continued weathering, biodegradation, and possible resuspension.
HOW OIL CHANGED
Crude oils contain thousands of compounds that, upon entering a marine environment, undergo significant compositional changes from weathering processes such as evaporation, dissolution, emulsification, dispersion, sedimentation/flocculation, microbial degradation, and photooxidation.
Most crude oil compounds are readily biodegradable and generally follow a clear degradation pattern: n-alkanes first followed by branched alkanes, lower molecular weight aromatics, higher molecular weight aromatics, and cyclic alkanes. Anaerobic biodegradation is a slower process than aerobic degradation, and crude oil compounds can remain relatively unaltered in reduced sediments and environments for long time periods and may appear as relatively fresh oil compared to surface oil exposed to aerobic conditions.
MICROBIAL RESPONSE AFFECTING OIL FATE
Macondo oil had a relatively low content of persistent resins and asphaltenes, and warm temperatures supported geochemical and biological degradation. The prevalence of oil-degrading bacteria generated a prompt response from the microbial community and subsequent biodegradation. Microbial communities in the plume were different from those in non-plume waters and exhibited a significant enrichment of hydrocarbon-degrading metabolic genes. Aerobic oxidation of short chain alkanes, propane, and butane caused up to 70% of oxygen depletion observed in the oil plume.
Residual oil trapped in Pensacola Beach sands showed a progression of microbial populations linked to hydrocarbon degradation. Early-responder microbes were followed by populations capable of aromatic hydrocarbon decomposition. Microbial abundance in oiled sands was 10-10,000 times that in clean sands in the first four months after oil came ashore. A typical beach-environment microbial community returned after one year but differed significantly from pre-spill communities.
DEEP OCEAN IMPACTS
Carbon from the spill was likely incorporated into the mesopelagic (200-1,000 m depth) food web through consumption of prey rich in depleted carbon. The nature of microbial communities in the deep sea likely changed. An 80-93% decline in benthic foraminifera was related to reducing conditions and increased polycyclic aromatic hydrocarbons (PAH) concentrations.
Deepsea megafauna had lower diversity and abundance near the spill site relative to regions farther away, though blue marlin, Atlantic sailfish, blackfin tuna, and dolphinfish showed no significant reduction in larval abundance. Bottom-dwelling golden tilefish had the highest concentrations of naphthalene metabolite levels in bile measured in fishes globally. Tunas and jacks collected near the spill site exhibited developmental crude oil cardiotoxicity, suggesting a possible loss of early predator recruits that spawn in open waters. Sperm whale acoustic activity decreased near the spill site by a factor of two and increased farther away, suggesting they relocated.
Hard-bottom communities, including natural and artificial reefs, suffered injuries that were severe and long-lasting. Macrofauna and meiofauna diversity had not recovered after four years, and community structure differences still persist. Deep-sea colonial corals, in particular octocorals near the spill site, showed visible evidence of impact, and flocculent material covering the coral contained chemical fingerprints associated with Macondo oil and DOSS (dioctyl sodium sulfosuccinate). Researchers returned to these coral eight times and observed continued impacts such as tissue death with some coral skeletons secondarily colonized by hydrozoans.
Field measurements showed that planktonic community abundance and species composition returned to pre-spill conditions within a year. Laboratory experiments indicated that zooplankton exposed to sublethal crude oil levels bioaccumulated five PAHs, which could increase their susceptibility to predation and enhance trophic transfer of toxic PAHs.
MARSH IMPACTS
There were immediate negative impacts in moderately to heavily oiled marshes in southeastern Louisiana. The average concentration of total alkanes and PAHs in June 2013 was 20 and 374 times pre-oiled conditions, respectively. Total alkane concentrations were on a trajectory to be near baseline levels by 2015, but this did not occur likely a result of multiple resuspension events from storms.
Some damaged marsh shorelines showed precipitous shoreline erosion at least 2.5 years after oiling due to damaged root systems. Marshes lost due to oiling and shoreline erosion will not return without human intervention. Forty-two months after the spill, heavily oiled marshes showed near-complete plant mortality, and live aboveground biomass was 50% of reference marshes. Decreased living marsh vegetation and population levels of some fauna were obvious for 2-5 years. Meiofauna density was lower along with S. alterniflora grasses in heavily oiled areas.
Fiddler crab average size declined and there were proportion shifts in two species composition. Periwinkle snails density declined, and a slow recovery in abundance and size distribution was related to habitat recovery. Worms, seed shrimp, and mud dragons had not recovered to background levels 48 months post-spill. Killifish showed little evidence of spill impacts. Horse fly abundance declined sharply. Arthropods were suppressed by 50% in 2010 but had largely recovered in 2011. Seaside Sparrow nests on unoiled sites were more likely to fledge than those on oiled sites. Loons varied in frequency with PAHs by year and exhibited reduced body mass as PAH concentrations increased.
These effects are expected to continue – possibly for decades – to some degree, or the marsh ecosystem will reach a new baseline condition in heavily damaged areas.
FISH & SEAFOOD IMPACTS
Commercial, recreational, and subsistence fisheries were closed in fall 2010 in areas where oil was observed and predicted to travel and reopened by April 2011. Impacts on fisheries productivity were relatively short-lived, with landings and their values returning to pre-spill levels or greater for most fishery species. However, long-term effects are yet to be determined. Laboratory studies indicate that early life stages of fish are generally more sensitive to oil and dispersant’s sublethal effects (with some resulting in reduced swimming performance and cardiac function) than adults.
Public health risks from exposure to crude oil residue through seafood or coastal beaches returned to pre-spill levels after the spill dissipated. Seafood from reopened areas was found to be safe for consumption, with PAH levels comparable to those found in common local processed foods. PAH concentrations detected in many seafood samples during and following the spill were at least 2 orders of magnitude below levels of public health concern. DOSS was detected in less than 1% of samples and at levels below public health concern.
Tests on SRBs showed that Vibrio vulnificus were 10 times higher than the surrounding sand and up to 100 times higher than seawater, suggesting that SRBs can act as reservoirs for bacteria including human pathogens. Coquina clams initially showed higher PAH levels relative to the surrounding sand, but levels decreased continuously and were undetectable in sand (one year) and Coquina tissues (two years).
DISPERSANT EFFECTS & FUTURE TECHNOLOGIES
Dispersant increased the oil fraction that spread within the water column and laterally displaced oil that reached the sea surface. Dispersants reduced droplet sizes and rise velocities, resulting in a more than tenfold increase in the downstream length of the surface oil footprint.
Chemical dispersants may be more toxic to some marine organisms than previously thought, and small oil droplets created by dispersant use and directly consumed by marine organisms are often more toxic than crude oil alone. Dispersant effects on microorganisms might be taxa-specific, and some studies suggest that dispersants stimulated biodegradation while others conclude the opposite. Degradation rates of hexadecane and naphthalene were more rapid in the absence of dispersants, as was the overall removal of the water-accommodated oil fraction.
Dispersant applied at the broken riser pipe helped form a deep water oil plume. DOSS was likely transferred to the plume and was later detected in surface sediments, on corals, and within oil-sand patties.
A future option is development of plant-based materials for efficient chemical herding of compact oil slicks into layers that are sufficiently thick to enable oil burning or skimming. Opportunities exist for new dispersants that work in synergy with current dispersants and mitigate some of their disadvantages. Examples include a system containing soybean lecithin and the surfactant Tween 80, substitution of lecithin for DOSS, and using carbon-based particles and silicas to stabilize emulsified droplets. Laboratory research needs to be conducted at concentrations and under conditions relevant to marine environments.
MODELING CAPABILITIES
Model improvements provide a better understanding of droplet formation in the turbulent plume above the wellhead. No model during the spill could predict droplet size distribution, which dictates rise times, dissolution, and biodegradation. Oil spill models now include the ability to simulate the rise of a buoyant oil plume from the seabed to the surface. Consideration of oil’s 3D movement permits the prediction of oil spreading through subsurface plumes. Our understanding of the near-surface oceanic layer and atmospheric boundary layer, including the influences of waves and wind, has also improved.
Oil spill modeling routines will likely be included in Earth system models, linking physical models with marine sediment and biogeochemical components. Advances in coupled nearfield-farfield dynamic modeling together with real-time, seven-day circulation forecasts allow for near-real-time tracking and forecasting of oil dynamics. This is the most promising approach for rapid evaluation of blowout predictions to support first response decisions.
* Overton, E.B., T.L. Wade, J.R. Radović, B.M. Meyer, M.S. Miles, and S.R. Larter. 2016. Chemical composition of Macondo and other crude oils and compositional alterations during oil spills. Oceanography 29(3):50–63
Socolofsky, S.A., E.E. Adams, C.B. Paris, and D. Yang. 2016. How do oil, gas, and water interact near a subsea blowout? Oceanography 29(3):64–75
Passow, U., and R.D. Hetland. 2016. What happened to all of the oil? Oceanography 29(3):88–95
Özgökmen, T.M., E.P. Chassignet, C.N. Dawson, D. Dukhovskoy, G. Jacobs, J. Ledwell, O. Garcia-Pineda, I.R. MacDonald, S.L. Morey, M.J. Olascoaga, A.C. Poje, M. Reed, and J. Skancke. 2016. Over what area did the oil and gas spread during the 2010 Deepwater Horizon oil spill? Oceanography 29(3):96–107
John, V., C. Arnosti, J. Field, E. Kujawinski, and A. McCormick. 2016. The role of dispersants in oil spill remediation: Fundamental concepts, rationale for use, fate, and transport issues. Oceanography 29(3):108–117
Passow, U., and K. Ziervogel. 2016. Marine snow sedimented oil released during the Deepwater Horizon spill. Oceanography 29(3):118–125
Tarr, M.A., P. Zito, E.B. Overton, G.M. Olson, P.L. Adhikari, and C.M. Reddy. 2016. Weathering of oil spilled in the marine environment. Oceanography 29(3):126–135
Joye, S.B., S. Kleindienst, J.A. Gilbert, K.M. Handley, P. Weisenhorn, W.A. Overholt, and J.E. Kostka. 2016. Responses of microbial communities to hydrocarbon exposures. Oceanography 29(3):136–149
Rabalais, N.N., and R.E. Turner. 2016. Effects of the Deepwater Horizon oil spill on coastal marshes and associated organisms. Oceanography 29(3):150–159
Murawski, S.A., J.W. Fleeger, W.F. Patterson III, C. Hu, K. Daly, I. Romero, and G.A. Toro-Farmer. 2016. How did the Deepwater Horizon oil spill affect coastal and continental shelf ecosystems of the Gulf of Mexico? Oceanography 29(3):160–173
Buskey, E.J., H.K. White, and A.J. Esbaugh. 2016. Impact of oil spills on marine life in the Gulf of Mexico: Effects on plankton, nekton, and deep-sea benthos. Oceanography 29(3):174–181
Fisher, C.R., P.A. Montagna, and T.T. Sutton. 2016. How did the Deepwater Horizon oil spill impact deep-sea ecosystems? Oceanography 29(3):182–195
Dickey, R., and M. Huettel. 2016. Seafood and beach safety in the aftermath of the Deepwater Horizon oil spill. Oceanography 29(3):196–203
************
The Gulf of Mexico Research Initiative (GoMRI) is a 10-year independent research program established to study the effect, and the potential associated impact, of hydrocarbon releases on the environment and public health, as well as to develop improved spill mitigation, oil detection, characterization and remediation technologies. An independent and academic 20-member Research Board makes the funding and research direction decisions to ensure the intellectual quality, effectiveness and academic independence of the GoMRI research. All research data, findings and publications will be made publicly available. The program was established through a $500 million financial commitment from BP. For more information, visit http://gulfresearchinitiative.org/.
© Copyright 2010- 2017 Gulf of Mexico Research Initiative (GoMRI) – All Rights Reserved. Redistribution is encouraged with acknowledgement to the Gulf of Mexico Research Initiative (GoMRI). Please credit images and/or videos as done in each article. Questions? Contact web-content editor Nilde “Maggie” Dannreuther, Northern Gulf Institute, Mississippi State University (maggied@ngi.msstate.edu).